Colorado Woman Joins Online Complaints About Birth Control Method
LOVELAND, Colo. (CBS4) Doctors tout Essure as an easy, non-invasive form of permanent birth control. But thousands of women are now complaining of complications that they say are related to the medical device. It's becoming an online movement to raise awareness about the problems.
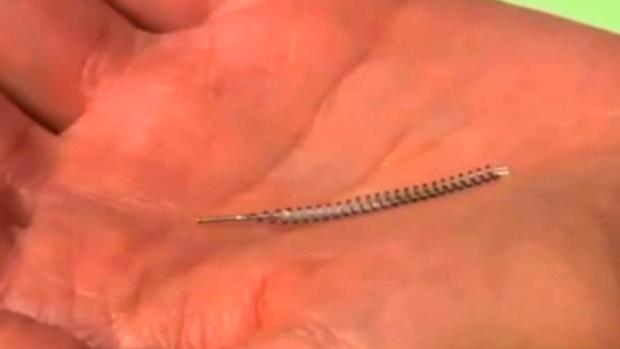
Essure is made Bayer Healthcare. It's a tiny coil that doctors insert into each of a women's fallopian tubes, within 3-months scar tissue forms, blocking the tubes and stopping further pregnancies.
LINK: Essure website
"We do the Essure procedure typically as an outpatient in the office, with only oral medications for pain and discomfort. And so over all I do believe it's a safer procedure," said Dr. Kara Alexandrovic, an OB/GYN with Metropolitan OB/GYN.
Alexandrovic has implanted Essure in as many as 50 patients. She's among hundreds of doctors across the country who offer the product and call it safe an effective. Alexandrovic says that Essure is the only form of permanent sterilization that does not require surgery under anesthesia, therefore, making it safer than other procedures.
"I've been very pleased with the results," Alexandrovic told CBS4.
Becky McGava Nelson is among the women who've posted their complaints about Essure online. She had the coils implanted 6-years ago when she and her husband were done having children.
"In my third year of having Essure, I passed out at a client's house. It was just the pain…it doubled me over," McGava Nelson told CBS4's Karen Leigh.
She complains of nausea, vomiting, dizziness, fever, and shaking, all of which she attributes to Essure.
"Pretty much 10-days…15-days out of the month, I was down," McGava Nelson added.
On September 4th, she had the coils removed.
"How do you feel now?" Leigh asked McGava Nelson.
"110-percent. I'm back to 30-days of life, instead of 15," she responded.
McGava Nelson posted her experience on a Facebook page about Essure. She's among thousands of women who've done the same. There's also a website started by consumer advocate Erin Brockovich that is collecting women's stories.
LINK: Erin Brockovich website
"I was actually very overwhelmed how quickly it built from 50 to a couple of hundred, to now thousands of stories," Brockovich said.
The Food and Drug Administration (FDA) approved Essure in 2002. The approval letter references company testing, and calls for further testing over 5-years. The company studies show a 99.8-percent effectiveness rate, better than other forms of permanent birth control. The studies track some complaints, like headaches, abdominal pain, back pain, but found they occur in less than 10-percent of patients.
RESOURCES: FDA Approval Letter
On October 29, 2013, the FDA issued a Review of Reported Problems. The federal agency acknowledges it's gotten 943 complaints about Essure. But the statement goes on to say that federal investigators reviewed the company-sponsored research and found no "new safety problems".
RESOURCES: FDA Review of Reported Problems
"At Bayer, we care about our patients and take the safety of our products very seriously," said Dr. Edio Zampaglione, a Vice President for Bayer Healthcare.
Zampaglione made that comment as part of a video response that Bayer Healthcare provided to CBS4 for this story, and gave no response to further questions about the product. A company statement says that approximately 750,000 women worldwide rely upon Essure for permanent birth control. And that the medical device has "a well-documented benefit-risk profile, with over 400 peer-reviewed publications and abstracts" supporting it's safety.
"Essure is one of the most effective contraceptives and has been on the market over 10 years," Zampaglione said on the video response.
RESOURCES: Written Statement from Bayer Healthcare
The FDA approval in 2002 extends preemption status to Essure, which means that it's exempt from state common laws that would allow injured patients to sue for damages. In other words, patients who suffer problems from Essure can't sue Bayer Healthcare under state law.
"They have no legal recourse ... which is very frustrating," Brockovich said.
Brockovich is using her website to gather signatures on a petition. She'd like 5,000 women to support her effort to lobby Congress to change the preemption law.
"There is a way to look at it, there is a way to recall it, it's just a matter of are we going to choose to recall it or not," Brockovich said.
"What do you want to do now? What is your goal?" Leigh asked McGava Nelson.
"I just want to reach out to as many girls as I can…whenever I can," McGava Nelson responded.
She'll continue to support the online campaign raising awareness about Essure.
--Written for cbsdenver.com by Special Projects Producer Libby Smith