CBS4 Health Specialist Kathy Walsh Volunteers For Novavax COVID Vaccine Clinical Trial
AURORA, Colo. (CBS4)- Coloradans can now volunteer for a major study of a new COVID-19 vaccine developed by Novavax.
I've reported on countless clinical trials and wondered what participating would be like. I decided what better time to find out then in the midst of a pandemic with the urgent need for vaccines.
On Tuesday, in a nondescript trailer in a parking lot on the Anschutz Medical Campus, nurse practitioner Jenelynn greeted me pleasantly.
"Thank you for coming in today, your participation in this trial," she said.
Then she got right to work.
"Do you have any comments, question or concerns?" she asked.
I volunteered for the phase 3 clinical trial of the COVID-19 vaccine developed by Novavax.
UCHealth is one of 115 study sites, mainly in the U S, working to determine its safety and effectiveness.
This was my first visit and it started with signing a number of forms. There were some easy screening questions.
"Are you of childbearing potential?" Jenelynn asked.
Nope. I then had a physical exam checking vital signs.
I moved on to research nurse, Trisha, who took 4 vials of blood and talked me through a COVID-19 test because I may need to do one at home.
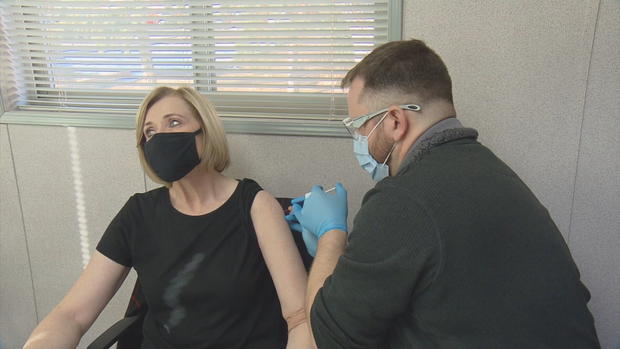
Eric, an intern pharmacist, arrived with the shot.
"Are you a fan of needles?" he asked.
Seriously, Eric?
I was given either the vaccine or a placebo, I don't know which and neither do the researchers.
With Novavax, two-thirds of the participants will get vaccine.
I was taught how to set up an electronic diary to record my temperature and health daily. I had to wait for 30 minutes to see if I had any adverse reactions to the shot.
I will go back for my next shot in 3 weeks. There are 8 to10 required visits over 26 months.
RELATED: Colorado Counties COVID Vaccine Distribution Information
The study literature says you may not benefit from being in this research, but I believe volunteering may benefit all of us in the future.
Anyone interested in participating can send an email to suzanne.fiorillo@cuanschutz.edu.