'It's Exciting': Denver Moderna COVID Vaccine Trial Participant Talks About Effective Rate
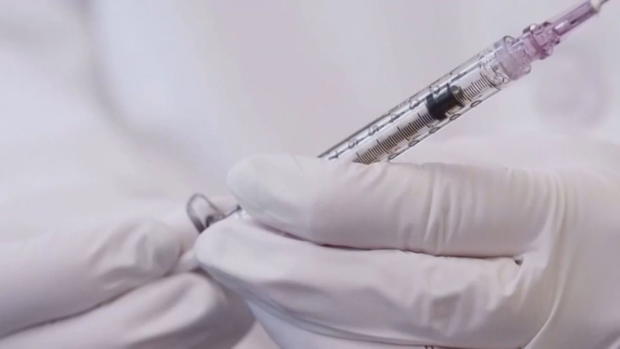
AURORA, Colo. (CBS4)– A second coronavirus vaccine in the U.S., that was tested on Colorado volunteers, is proving to be highly effective in the trials. Both participants and doctors at UCHealth are optimistic about the results shared on Monday. Moderna said its vaccine appears to be 94.5% effective according to preliminary data from the company.
"I think it's huge, it's so exciting to be a part of this and to know all these other people are taking some sort of risk," said Jonathan Mendez, a participant in the trial who lives in Denver. "I think this is amazing, last week I was impressed by the Pfizer vaccine being at 90%, for this one to be even better."
Mendez took his first shot five weeks ago and then the second dose in the past week as a part of the 25-month study. He has to record his temperature daily and report any symptoms or side effects he gets along the way. He tries to follow the standard guidelines for limiting the spread of COVID-19 by sticking to a small group of friends and avoiding dining-in at restaurants.
"We have to get this right, I don't think that we're rushing too quickly. We should be a little hesitant at what comes out," he told CBS4. "The rigor by which this vaccine is being developed, is to a very high standard that I think most people should feel confident and trust the experts on this."
UCHealth is one of 89 research sites across America that participated in phase three study trials of the Moderna vaccine. It was just last week when competitor Pfizer Inc. announced its own COVID-19 vaccine that boasted a 90% effective rate. Both vaccines require people to receive two shots several weeks apart. The results are "truly striking," said Dr. Anthony Fauci, the U.S. government's top infectious diseases expert, who earlier this year said he would be happy with a COVID-19 vaccine that was 60% effective.
"We have two independent scientific experiments that come up with the same results that demonstrate that both of these vaccines are highly effective," said Dr. Thomas Campbell, a professor of infectious disease medicine at CU Anschutz Medical Campus. "We are able to do that already with lots of other vaccines, I don't think that will be a big issue."
Campbell says the two vaccines have similar technology and strategy as mRNA treatments but cautions these are early results from studies that began in July. Both trials will span two years so the effectiveness and safety can be examined for a longer period of time.
If the FDA approves emergency use of Moderna's or Pfizer's vaccine there is expected to be limited supplies before the end of the year. Those considered high risk will be among the first to receive the vaccine.
"We still have a lot of work to do, continuing to follow our volunteers who have joined this important study," Campbell told CBS4 on a video conference call. "It's huge, it's very welcome news given what's going on with COVID across the country."
The road ahead for a vaccine includes manufacturing, distribution, and administration. While both drug companies expect to have at least 20 million doses by the end of the year, it will only be a fraction of all that is needed in just the U.S. Given that each vaccine requires two does per person, the amount needed is much larger than current production estimates. But Campbell also wants to see confidence in the public so people will take the vaccine when it is ready.
While both vaccines need to be stored in cold temperatures, unlike the Pfizer option, Moderna does not need to be stored in ultra-low freezing temperatures. Standard refrigeration will hold the Moderna vaccine for around a month. The resources needed for that type of storage should be at most hospitals at clinics. This will not change the timeline for the general public to receive the vaccine, Campbell estimates that will be the second quarter of next year.
Gov. Jared Polis has stated previously that the vaccine would be distributed in three phases in Colorado with those in the high-risk category receiving the vaccine first, followed by the population that falls into the other two phases. Last week, Polis said that that goal is to have enough doses so that everyone in Colorado can be vaccinated if they choose, by late spring 2021.
"It gives us hope that even though we're going to have some tough months ahead of us coming into December and January with COVID and many people are going still be getting sick, beyond that," Campbell said. "There's hope that we'll finally be able to get COVID under control in the United States."
One of the challenges is that both Moderna and Pfizer vaccines are frozen but at different temperatures. Moderna's vaccine can last longer in a refrigerator than initially thought once thawed, up to 30 days. Pfizer's vaccine requires long-term storage at below zero temperatures.
"I think this is very exciting, I am very hopeful for the future, and along with a lot of these social distancing measures," Mendez said. "This is what we need to open and for life to sort of go back to a semblance of normalcy."